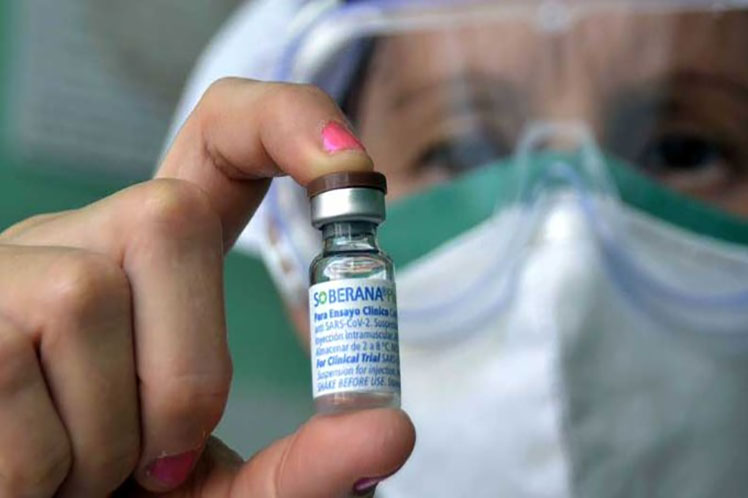
Havana, Sep 27 (RHC) On Monday, the Finlay Institute of Vaccines (IFV) highlighted the application of doses of the Soberana Centro clinical trial against Covid-19, in the province of Cienfuegos.
The scientific entity wrote a message on its account on the social network Twitter in which it reports the conclusion in that territory of the application of the third and last dose of the vaccine in more than 1,160 subjects.
It detailed that the immune response of Soberana 01 was evaluated with respect to two of Soberana 02 plus Soberana Plus.
Recently, Dr. Yanet Climens of the Finlay Institute of Vaccines collective clarified that the study has not yet concluded because the vaccinated subjects will be followed up every four months, a process that will be extended for a year.
The third dose (Soberana Plus) was applied to each of the participants, which gave continuity to the previous inoculations of Soberana 01 and Soberana 02 in a blind study.
The Soberana Centro clinical trial seeks to evaluate the immunogenicity of the prophylactic anti-SARS-CoV-2 vaccine candidate FINLAY-FR-1 (Soberana01) with respect to FINLAY-FR-2 (Soberana02), both in a heterologous scheme with FINLAY-FR-1A (SoberanaPlus).
The expert said that at the end of the research it is expected that the effectiveness of Soberana 01 will be equal or surpass Soberana 02. (Source: PL)

